Effecting Synergistic Mechanisms of Action to Attack Hard-to-Treat Cancers
Our lead candidate, DPX-Survivac, is a T cell activating immunotherapy that combines the utility of the DPX platform with a novel target: survivin.
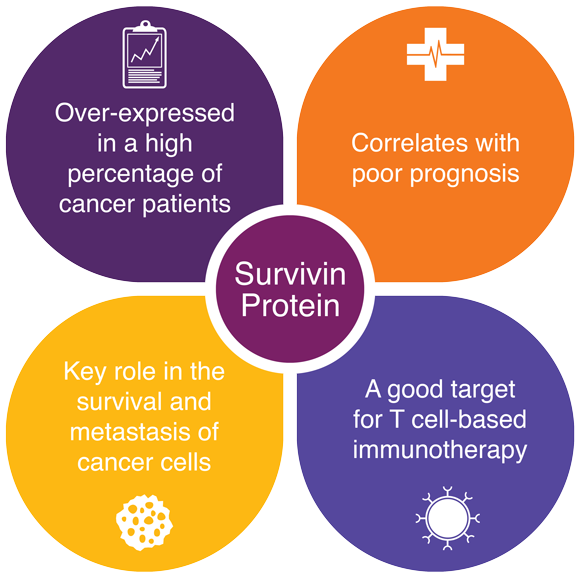
Survivin, a cancer stem cell antigen and biomarker of cancer progression, is a particularly valuable target because it has the capacity to identify and disrupt cancer biology on multiple levels:
- It is cancer-specific but broadly expressed, currently associated with over 20 types of solid and hematological cancers.
- It is actively implicated in the dysregulation of cell proliferation and cell death.
- It is associated with particularly aggressive tumors and resistant to chemotherapeutic agents.
Survivin isn’t just a tumor-associated-antigen (TAA), it is also an instigator of the disease itself. By infiltrating this target, not only do we efficiently seek out cancer-causing cells while leaving healthy tissues unaffected, but we can also disrupt the fundamental processes of cancer cell production and survival.
IMV has an exclusive, worldwide license to survivin from Merck KGaA.
DPX-Survivac in combination with metronomic cyclophosphamide hits the mark on several characteristics of an optimal immunotherapy approach. It has the capacity to:
- TARGET a cancer-specific marker
- IMPACT tumor progression disabling key tumor survival mechanisms
- Be POTENT enough to address even the hardest-to-treat of cancers
- SUSTAIN exposure to ensure the development of immune memory and prolonged tumor death
Multiple Markets & Broad Opportunity
Because we believe that DPX-Survivac has significant market potential across multiple indications, we have pursued a very deliberate, focused clinical strategy where we think it can add the most value. Building on preclinical research that indicated our anti-cancer therapies are able to induce T cell responses, we began pursuing therapies for DPX-Survivac where we believe it can most directly benefit underserved patients with hard-to-treat cancers, particularly those who have not responded to next-generation immuno-modulator therapies. Our primary initial indication for DPX-Survivac is in advanced ovarian cancer.